How Do ADC Services Accelerate Antibody-Drug Conjugate Development?
ADC services help biotech teams move antibody-drug conjugates from idea to clinic with greater speed and control. These services combine antibody engineering, conjugation science, analytics, and development strategy. By centralizing expertise and workflows, ADC services remove bottlenecks, reduce trial and error, and allow developers to focus on decisions that drive faster, more predictable progress across complex development programs globally.
The Role of ADC Services in Early-Stage Development
Antibody Selection and Engineering Efficiency
ADC services accelerate early development by streamlining antibody selection and engineering. Teams rapidly screen targets, optimize binding, and adjust formats for conjugation compatibility. Clear ownership of these steps reduces iteration cycles. As a result, biotech developers can advance stronger antibody candidates sooner, lowering rework risk and improving alignment between biological performance and downstream chemistry and manufacturing requirements from the start phases.
Payload and Linker Evaluation Strategies
Payload and linker evaluation requires balancing potency, stability, and safety. ADC services apply structured testing to compare mechanisms, linker chemistries, and release behavior. This focused strategy prevents over experimentation. By narrowing options quickly, biotech teams gain confidence in design choices, shorten discovery timelines, and reduce late stage surprises caused by unsuitable payload strength or unstable linker behavior in complex biological systems.
Rapid Feasibility and Proof-of-Concept Studies
ADC services enable rapid feasibility and proof of concept studies by coordinating materials, assays, and data analysis. Providers generate early conjugates, test activity, and assess developability in parallel. This approach helps biotech teams confirm viability fast, stop weak programs early, and redirect resources efficiently. Faster feasibility decisions protect budgets and accelerate promising ADC candidates toward structured development plans and internal milestone commitments.
Process Development Support from ADC Services
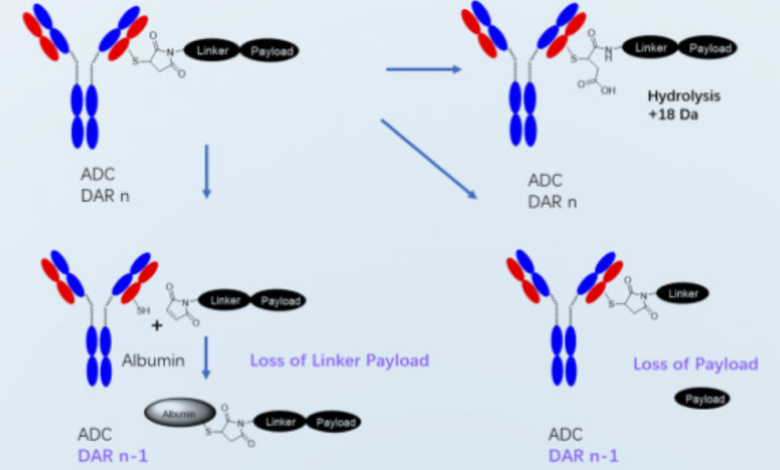
Optimizing Conjugation Methods and DAR Control
Optimized conjugation methods are critical for speed and consistency. ADC services refine reaction conditions, control to drug-to-antibody ratio, and reduce heterogeneity. Experienced teams anticipate common failure points and adjust processes early. This expertise minimizes repeated experiments, ensures reproducible outputs, and helps biotech developers establish reliable foundations for scale up without sacrificing biological activity or analytical clarity during development progression phases.
Improving Stability and Manufacturability Early
ADC services focus on stability and manufacturability during early process development. Teams assess aggregation, degradation, and storage behavior while designs remain flexible. Early fixes prevent costly redesigns later. By addressing manufacturability risks upfront, biotech companies shorten timelines, simplify regulatory paths, and improve confidence that selected ADC candidates can move smoothly from laboratory batches to clinical supply without unexpected setbacks.
Establishing Reproducible and Scalable Processes
Reproducibility supports speed across development stages. ADC services establish scalable processes that perform consistently across batches. Clear parameters, defined controls, and documented methods reduce variability. This preparation allows biotech teams to transition faster into GMP environments, support clinical timelines, and avoid delays caused by process instability or incomplete knowledge transfer when programs expand beyond early research settings.
Analytical and Preclinical Capabilities Within ADC Services
Comprehensive ADC Characterization and Testing
ADC services provide comprehensive characterization using advanced analytical tools. Teams assess identity, purity, drug loading, and structural integrity. Rapid access to these results supports quick decision making. By integrating analytics into development workflows, biotech companies identify issues early, reduce repeat studies, and maintain momentum without waiting for external testing cycles that often slow complex biologic programs.
Supporting In Vivo and In Vitro Preclinical Studies
Preclinical support within ADC services aligns in vitro and in vivo studies with development goals. Providers generate material, design studies, and interpret results efficiently. This coordination shortens study timelines and improves data relevance. Biotech teams gain actionable insights faster, allowing them to adjust designs or progress candidates confidently toward regulatory enabling studies without fragmented vendor management or misaligned experimental execution.
Data Integration for Informed Development Decisions
ADC services integrate data across chemistry, biology, and analytics into unified views. Clear reporting highlights trends, risks, and trade offs. This transparency helps biotech leaders make faster, better decisions. When data flows smoothly between functions, teams avoid delays caused by data gaps, miscommunication, or repeated analysis, enabling consistent progress toward defined development milestones and investment readiness checkpoints.
How ADC Services Reduce Timelines and Development Risk
Parallel Workflows and Integrated Development Models
ADC services reduce timelines by running tasks in parallel rather than sequentially. Integrated models allow discovery, process development, and analytics to progress together. This structure compresses schedules and avoids waiting between steps. For biotech companies, parallel workflows increase efficiency, reveal issues earlier, and create momentum that supports faster advancement toward clinical entry and competitive positioning within crowded therapeutic areas.
Avoiding Common ADC Development Pitfalls
Many ADC programs slow down due to predictable pitfalls, including unstable conjugates or unscalable processes. ADC services apply prior experience to avoid these issues. Teams recognize warning signs early and apply proven solutions. This proactive approach saves time, reduces wasted investment, and protects biotech companies from delays that often arise when complex development challenges appear late in the program lifecycle.
Preparing for IND and Clinical Transition Faster
ADC services support faster IND preparation by aligning development activities with regulatory expectations early. Documentation, data packages, and process understanding evolve together. This readiness reduces last minute gaps and review delays. Biotech teams benefit from smoother clinical transitions, stronger submissions, and shorter paths from preclinical success to first in human studies under increasing competitive and funding pressures.
Conclusion
ADC services accelerate antibody-drug conjugate development by combining expertise, integration, and execution speed. They reduce uncertainty, compress timelines, and support better decisions at every stage. For biotech companies facing limited resources and high complexity, these services provide structure and momentum. Strategic use of adc services can be the difference between stalled programs and successful clinical advancement.





